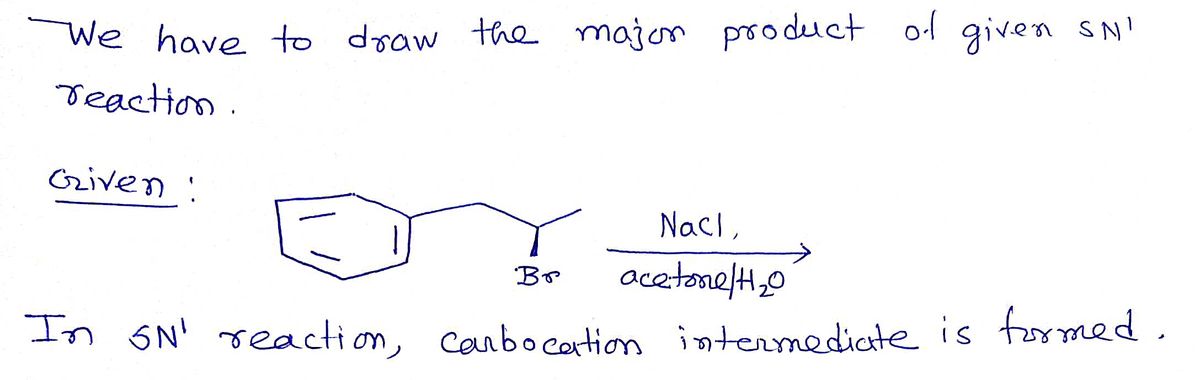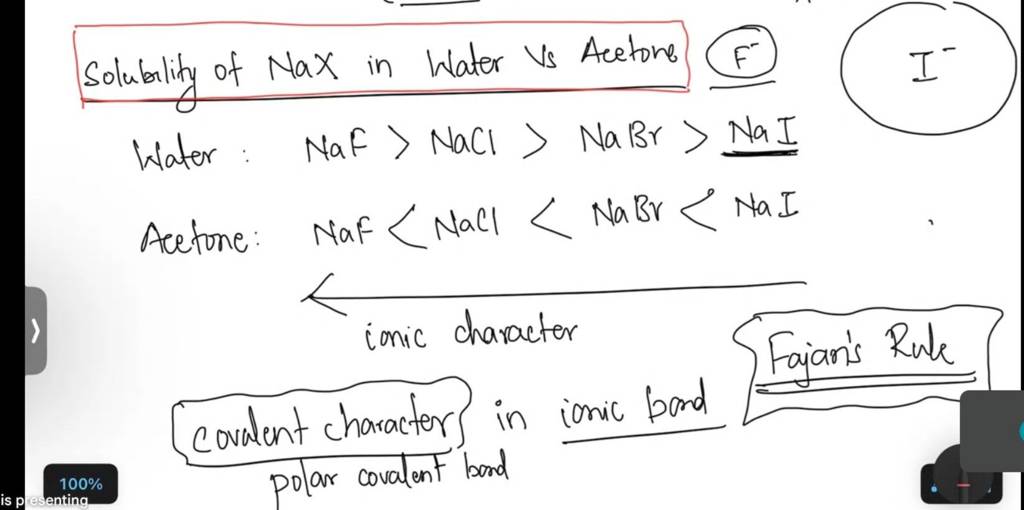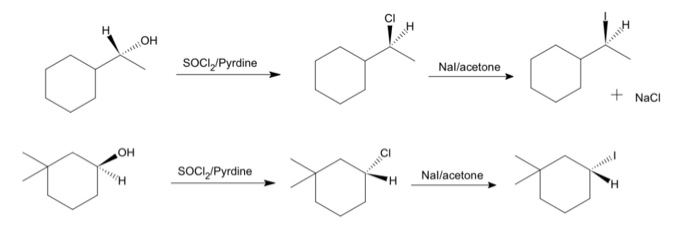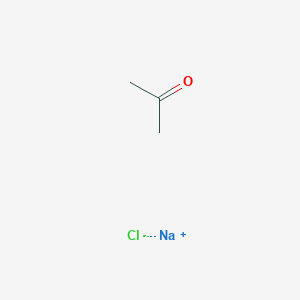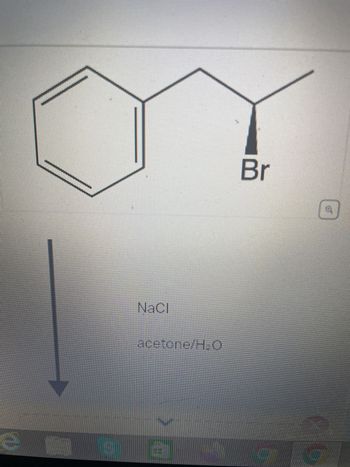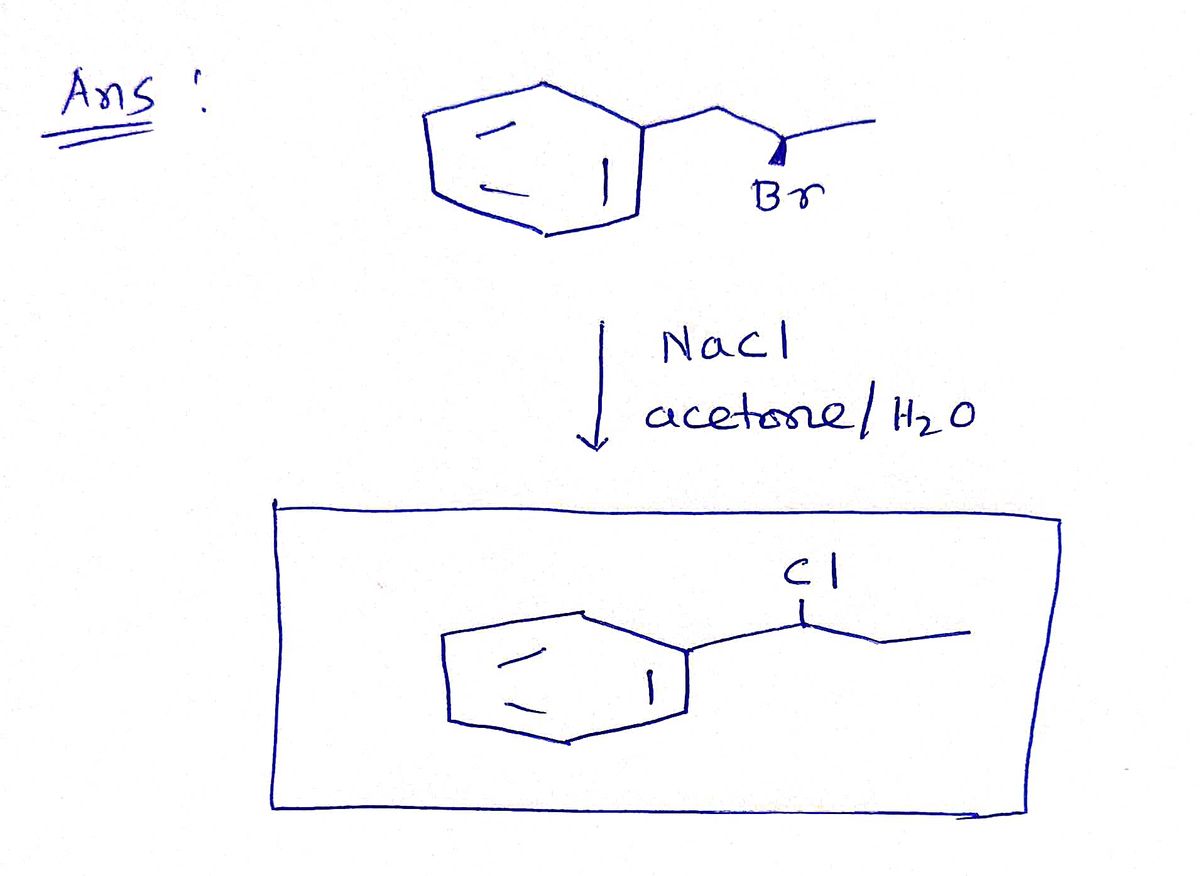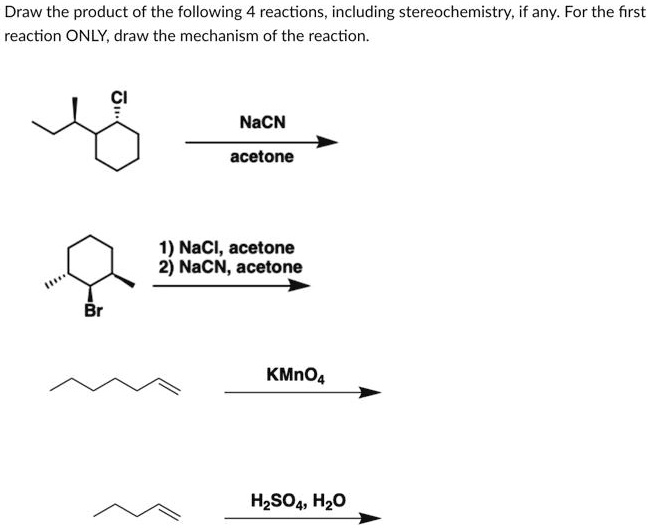
SOLVED: Draw the product of the following reactions, including stereochemistry, if any. For the first reaction ONLY, draw the mechanism of the reaction: 1) NaCl, acetone 2) NaCN, acetone KMnO4 H2SO4, H2O
Radiolysis-Induced Crystallization of Sodium Chloride in Acetone by Electron Beam Irradiation | Microscopy and Microanalysis | Cambridge Core

Ultra-high response acetone gas sensor based on ZnFe2O4 pleated hollow microspheres prepared by green NaCl template - ScienceDirect

SOLVED: 1. Sodium iodide in acetone: Nal readily dissolves in acetone: The moderately polar aprotic solvent promotes Sn? reactions lodide is an excellent nucleophile: The solvent does not favor ionization of alkyl
![PDF] Modeling vapor-liquid-liquid-solid equilibrium for acetone-water-salt system | Semantic Scholar PDF] Modeling vapor-liquid-liquid-solid equilibrium for acetone-water-salt system | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b36fc3bac22e494bb53655c2d2890a2857600723/7-Figure5-1.png)
PDF] Modeling vapor-liquid-liquid-solid equilibrium for acetone-water-salt system | Semantic Scholar
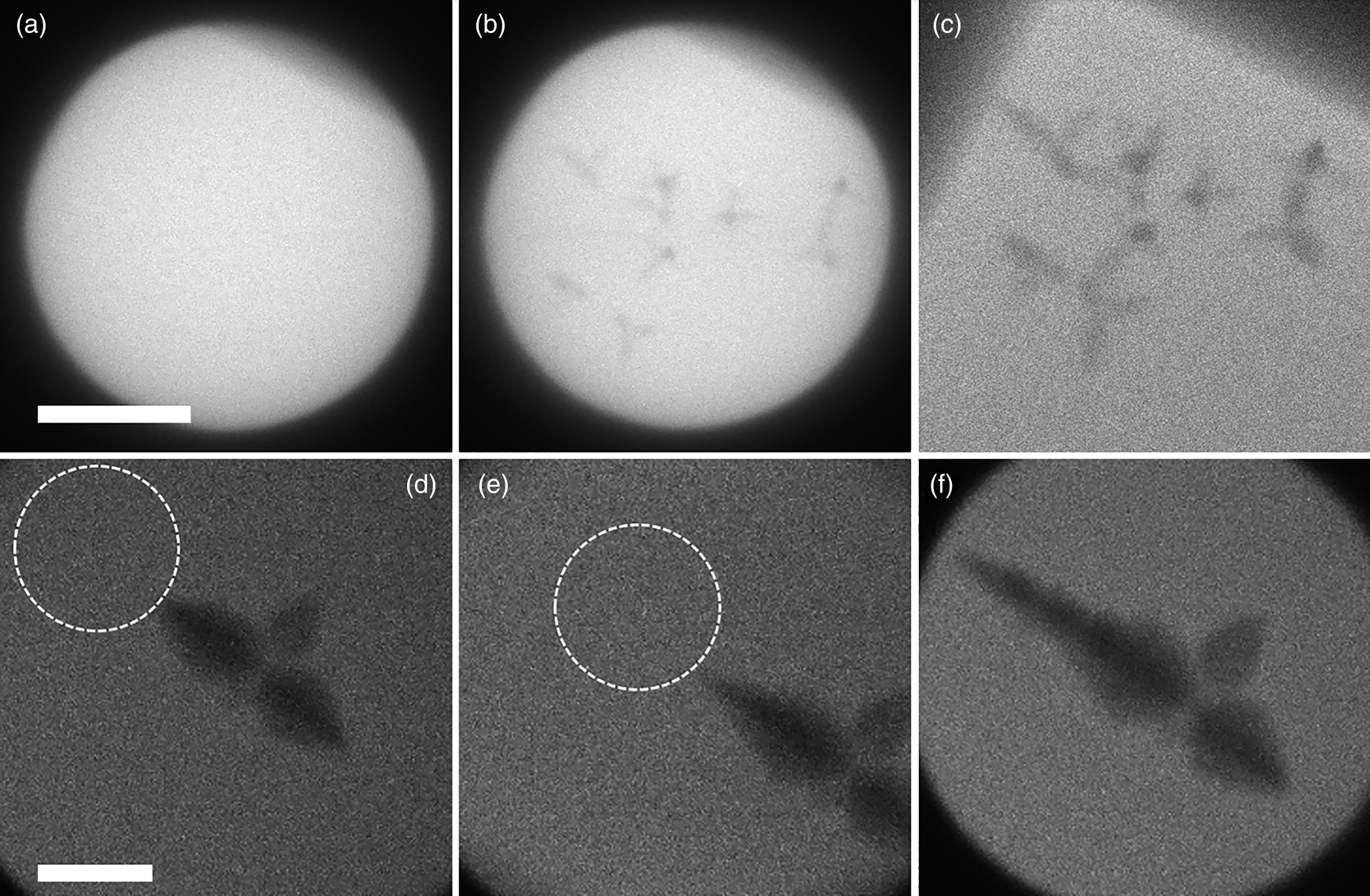
Radiolysis-Induced Crystallization of Sodium Chloride in Acetone by Electron Beam Irradiation | Microscopy and Microanalysis | Cambridge Core
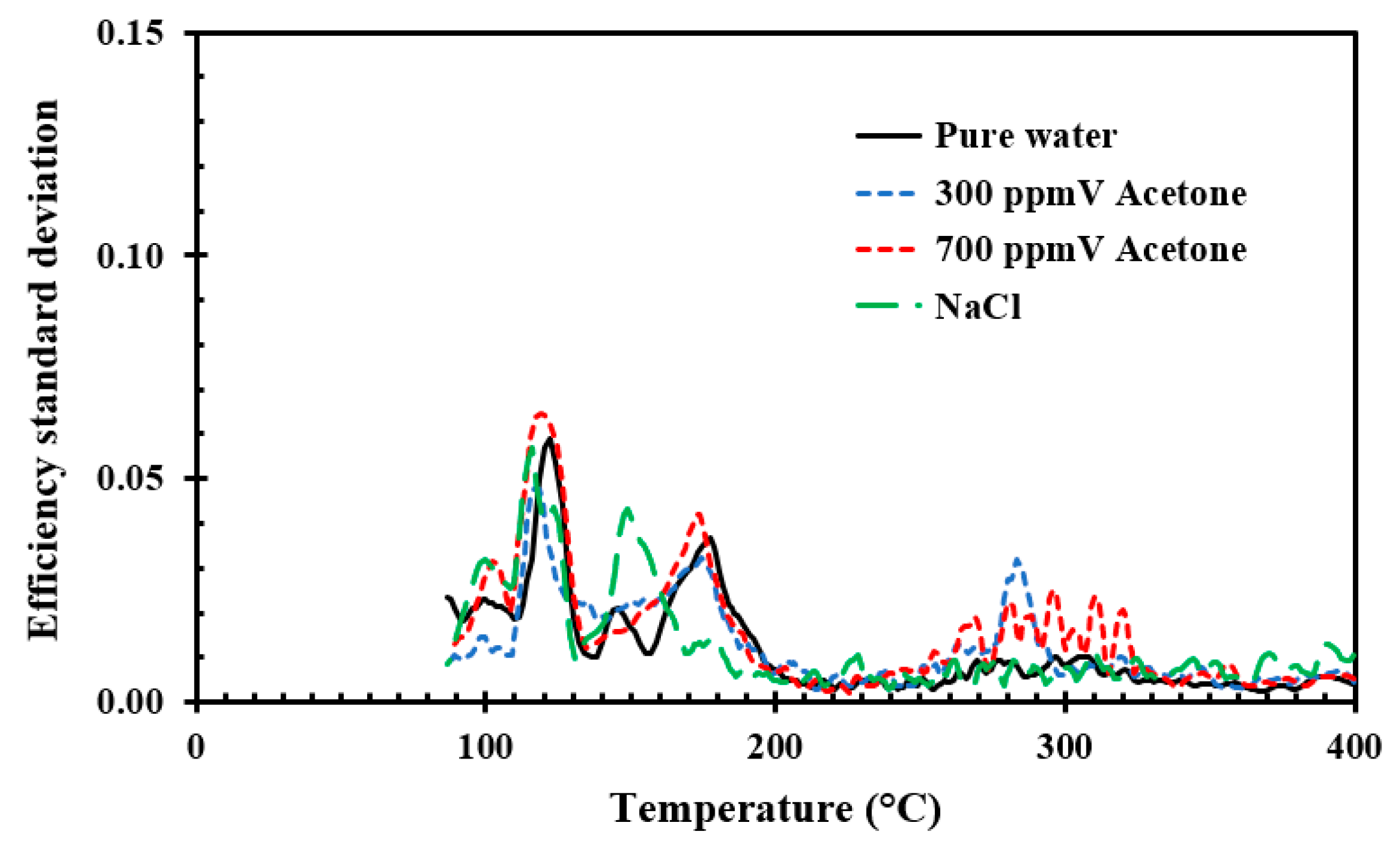
Energies | Free Full-Text | Influence of Acetone and Sodium Chloride Additives on Cooling Efficiency of Water Droplets Impinging onto Hot Metal Surfaces

PDF) Salting-out effect of single salts NaCl and KCl on the LLE of the systems (water + toluene + acetone), (water + cyclohexane + 2-propanol) and (water + xylene + methanol) | A. Hasseine - Academia.edu

NaCl recovery from organic pollutants-containing salt waste via dual effects of aqueous two-phase systems (ATPS) and crystal regulation with acetone - ScienceDirect

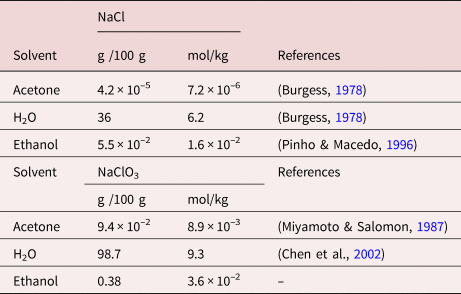
Solubilities of NaCl, KCl, LiCl, and LiBr in Methanol, Ethanol, Acetone, and Mixed Solvents and Correlation Using the LIQUAC Model | Industrial & Engineering Chemistry Research
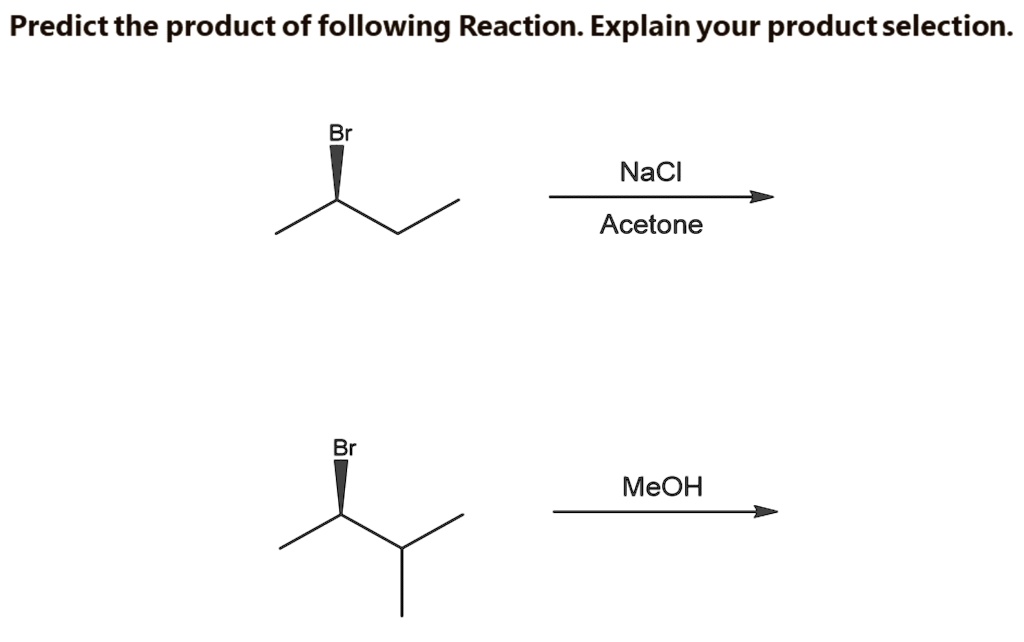
SOLVED: Predict the product of following Reaction. Explain your product selection: NaCl Acetone MeOH